Stainless steel protects itself
Download Pdf
Unlike other metals that require paints or external coatings to resist corrosion, stainless steel has an intrinsic protection mechanism. Its resistance does not depend on a layer applied afterwards, but on a metallurgical property inherent to the material itself.
The key: the chromium oxide passive layer
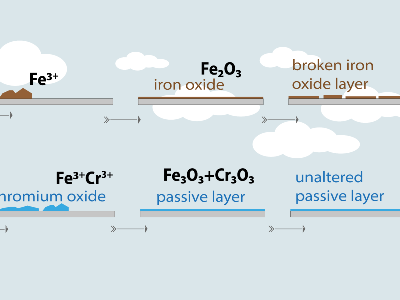
To be considered “stainless,” steel must contain at least 10.5% chromium. When this element interacts with oxygen in the air or water, it spontaneously forms a nanometric film of chromium oxide on the surface.
Technically, this film is the result of a controlled and beneficial oxidation process which, unlike common rust (iron oxide), prevents corrosion from progressing into the interior of the metal. This layer has several fundamental characteristics:
- Nanometric thickness: It is invisible and hermetically seals the surface.
- High chemical stability: It acts as a physical and electrochemical barrier that blocks the reactivity of iron. As a passive process, protection occurs naturally without external intervention.
- Complete adhesion: The layer is firmly bonded to the metallic substrate. This not only prevents it from flaking off, but also allows it to regenerate almost instantaneously in the event of mechanical damage such as scratches or cuts.
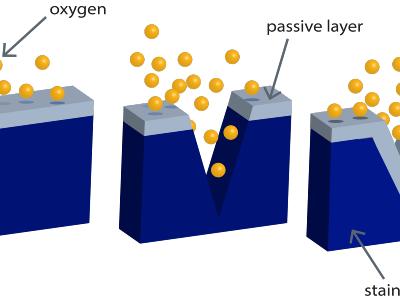
The effectiveness of this protection fundamentally depends on the availability of oxygen. In environments where air does not circulate or under accumulated dirt deposits, the passive layer has difficulty reforming. Therefore, the key to maximizing the service life of stainless steel is not to isolate it, but to keep its surface clean so that its self-healing nature can preserve the material’s integrity indefinitely.